Hand-Cracked, Wind-Up Heart Rate Monitor for Diagnosing Complications in Fetus’ Heart
Through a partnership between global tech firm, Royal Philips and South Africa-based non-profit organization PowerFree Education Technology (PET), a revolutionary and inexpensive medical technology breakthrough for diagnosing fetal heart complications during labor has been developed.
Dubbed the Wind-up Doppler Ultrasound Fetal Heart Rate Monitor, this is a medical technology device developed to curb the high infant mortality rate that results from fetal heart complications during labor. Although the technology, is still in its beta form, the two tech firms are working on developing it further and have it commercially applicable in the medical fields.
The ultrasound device works by accurately counting the fetal heart rate during child labor and promises to prevent infant deaths and damage to the infant’s brain during delivery. The device helps the midwife or the delivering nurse detect if or when the infant’s heart rate is slowing down, which is a likely indicating that the fetus is not getting enough oxygen supply.
“Current methods to measure the fetal heart rate are either too expensive, too inaccurate or rely on replaceable batteries or electricity to run. The Wind-up Fetal Doppler is especially designed to empower midwives and delivering nurses to give better care.” – The Company said.
The technology is being developed with semi-urban and rural areas in Africa being in mind; where often women and infant die as a result of easily preventable complication during the process of child bearing.
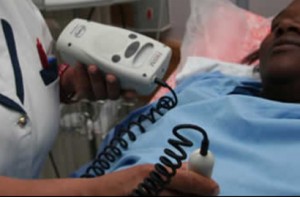

Philips African Innovation Hub, the self-proclaimed center for developing innovations “in Africa-for Africa”, has released the first prototype of the wind-up Doppler. But before this medical tech device goes fully into the general medical practice, it first must be subjected to clinical testing and regulatory approval.
“We are very excited about the collaboration with Philips,” said, Dr Francois Bonnici, the Director of PET and the Director of the Bertha Centre for Social Innovation at the University of Cape Town. Dr Bonnici further said he hoped that the partnership between PET and Royal Philips would make the “innovation available and affordable for frontline health care workers across the African continent.”
Article Source >> Royal Philips.




